Abstract
Background
The biosimilar rituximab (Redditux) was approved in Turkey for all indications of the reference molecule (MabThera) in March 2018. Large clinical trials and real-life experiences are lacking in hematological malignancies.
Aims
We aimed to evaluate the efficacy and safety of Redditux in de-novo diffuse large B-cell lymphoma (DLBCL).
Methods
Our institution decided to provide Redditux for hematological indications since February 2019. We retrospectively analyzed medical records of 51 consecutive de-novo DLBCL patients (pts) diagnosed between February 2019 and September 2019 in the hematology department of Istanbul University Istanbul Medical Faculty. We compared the response rates with historical controls treated with MabThera-CHOP at Cerrahpaşa Medical Faculty. Our study was approved by I.U. Istanbul Medical Faculty Ethical Committee (2019/1454).
Results
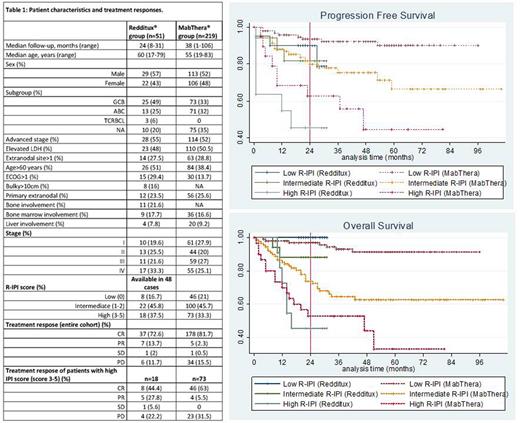
A total of 51 pts without CNS involvement received Redditux-CHOP. Median follow-up was 24 months (range: 8-31). A median of 6 cycles of biosimilar (range: 4-8) was administered. Four pts with high CNS-IPI score received four intrathecal methotrexate injections and 13 pts had additional radiotherapy for their initial bulky disease. The patient characteristics and response rates of the Redditux and historical MabThera cohorts are summarized in Table 1.
Apart from 6 cases who were refractory to Redditux-CHOP, 8 pts had progressive disease (6 with CR, 1 with PR and 1with SD) in the follow-up. The median time to relapse was 11.5 months for 6 cases who had CR following first-line treatment. Five of 8 cases with PD experienced CNS relapse. Their CNS-IPI score (Schmitz et al. J Clin Oncol 2016) were low in 2 pts, intermediate in 2 pts and high in 1 patient. Of 11 pts with bone involvement at the time of diagnosis, three cases had CNS relapse (p=0.028). Two pts with CNS relapse were treated with intrathecal chemotherapy only due to their poor performance status. Eight pts received salvage combination chemotherapy [R-ICE (n=3); R-benda (n=3); R-DHAP (n=1); MATRix (n=1)] and two of them responded. One of these 2 cases underwent auto-SCT and the other proceeded to allo-SCT; however, he died during conditioning treatment.
Ten pts died in the follow-up. Causes of death were progressive disease (n=7, two cases with CNS involvement), infection during allo-conditioning (n=1), post-COVID herpes zoster infection (n=1) and unknown (n=1).
The 24 month PFS and OS rates were 75.8% (95% CI: 0.61-0.85) and 80.3% (95% CI: 0.67-0.89) for Redditux cohort, respectively. In the historical MabThera group, the 24 month PFS and OS rates were 85.2 (95% CI: 0.79-0.90) and 81.4% (95% CI: 0.75-0.86), respectively.
For pts with high R-IPI score in the Redditux cohort (3-5); the 24-month PFS and OS rates were 54.2% (95% CI: 0.29-0.74) and 55.6% (95% CI: 0.31-0.75), respectively. In the historical Mabthera group, the 24-month PFS and OS rates were 68.7% (95% CI: 0.53-0.80) and 59.4% (95% CI: 0.47-0.70), respectively. Although the PFS rates seems to be worse in high R-IPI cases receiving Redditux, the difference was not significant (p=0.18; Figure 1).
AEs were reported in 51% (n=26) of patients. Most common AE was grade 2 infusion reactions (shivering, nausea, fever) requiring medical intervention in 20% of pts, accompanied with rash in half of them. Grade 3&4 AEs were leucopenia (n=2; 4%), neutropenia (n=20; 39%) febrile in 2 cases, anemia (n=6; 12%), thrombocytopenia (n=3; 6%). Grade 2 pneumonia (n=2) and urinary tract infections (n=2) were other infectious complications.
Conclusion
Although the PFS rate at 24 months in high-IPI group treated with Redditux seems to be lower compared to MabThera treated historical control group, survival rates were not significantly different. Our results should be cautiously evaluated due to small sample size. Compared to the original trial of MabThera added to CHOP based regimen, (Coiffier et al N Eng J Med. 2002), our CR rates in stage 2-4 pts seem to be slightly lower (70.7% vs 76%), although the OS rates are quite similar (86.3% vs 82%). Grade 3&4 neutropenia requiring empirical administration of G-CSF was 39% in our cohort. Infusion reactions were observed in 20% of pts, which was reported to be around 30% with original molecule (Patel et al. Clin Lymphoma Myeloma Leuk 2019). The CNS relapse rate was relatively high (9.8%) in our cohort. Prospective randomized clinical trials are needed to determine the efficacy and safety profile of Redditux.
Ferhanoglu: Takeda Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Roche: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees.